 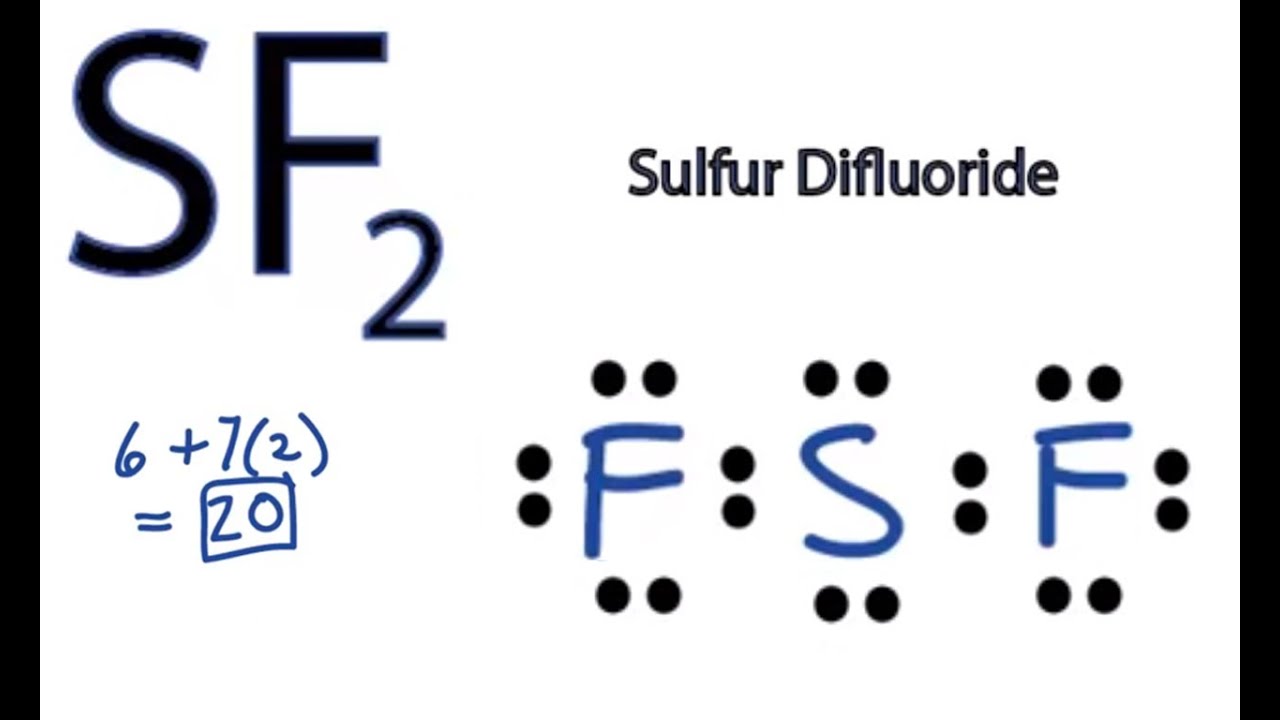 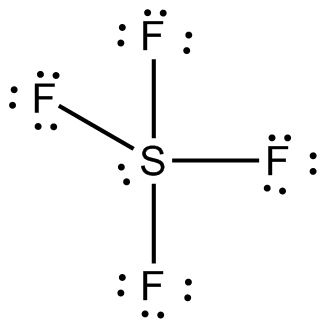
Case ba(oh)2 ofe2o3 csbr br25 how many valence electrons in the following chemical species in its lewis structure?ĭraw Lewis Structure For Bef2 from s3. The two lone pairs of electrons push the fluorine atoms downwards due to the repulsive forces, and as a result, the shape of this molecule is bent. 
Deduce the molecular polarity of cisplatin. Boron trifluoride is a versatile lewis acid that forms adducts with such lewis bases as fluoride and ethers: Lewis structure, then the molecular geometry of the molecules. Hydrogen is usually surrounded by 4 electrons in a valid lewis structure. 
If you draw a trial structure in which br is the central atom and every atom has an octet, you will have used 20 electrons (2 lone pair on br). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
